An important update is available for FreeStyle LibreLinkØ. Check here for more information.
 Menu
Menu
27 Sep 2018
Biological controls are largely self-explanatory as these should always be considered, regardless of the type of experiment. In the context of a flow cytometry experiment, controls to consider could be:
Compensation controls are always required if “spillover”, or spectral overlap between two fluorophores is likely.
Decide what type of compensation controls, either cells, beads or both, are appropriate for your experiment. If using beads, do remember to check the species the antibodies are raised in, as many antibody capture beads are species-specific.
Things to keep in mind:
In most cases, core facility staff will be monitoring your facility’s instruments to ensure there are no technical issues and that day-to-day variability is kept to a minimum. This is usually done by running an automated quality control check with fluorescent microspheres (beads). In addition, further steps can be taken by the researcher to reduce variability from experiment to experiment. On BD instruments, application settings can be stored so that any slight day-to-day variation in voltages is automatically adjusted for. Using application settings is highly encouraged, especially for longitudinal studies such as clinical samples.
In addition, users can always set their own instrument control by using a fluorescent bead to set a target MFI (after optimal voltages have been set), and running this prior to each experiment to ensure/track instrument stability over time. This helps flag any technical issues and adds “peace of mind” that all experiments will be comparable when collected over an extended time period. This can also be done as a quick and easy step to check cleanliness between users when instruments are heavily booked.
FMO controls are samples that contain all the antibodies in a panel, minus one of them. These are important to assess panel design and control for fluorescence spillover. Compensation cannot “fix” all undesirable affects caused by using a large number of fluorescent reagents. Therefore, FMO controls can help reveal patterns or combinations that should be changed or avoided. FMO controls should be done for all the fluorophores in your panel when first setting up and optimising a multicolour experiment. Once optimised, FMOs should not necessarily be required for every reagent in a panel. In fact, very few FMO controls should be required if careful panel design is performed.
For example, if an FMO control reveals that a bright fluorophore affects resolution of a dim marker, panel design should be reassessed. An FMO control cannot correct for this undesirable affect, it just reveals it. The main use of an FMO control in a fully optimised, well executed panel should be to accurately gate to identify a positive from a negative population when the expression levels are low or poorly resolved due to the biology, not poor reagent choice.
FMO’s can also be useful to help define background or negative staining when samples contain auto fluorescent populations. When cells have a significant inherent level of autofluorescence, these populations can sometimes appear positive, so it is a good idea to first confirm auto signal in every channel to avoid the potential for false positives.
Therefore, FMO controls are important for evaluating multicolour panel iterations as they reveal:
This means that when assessing FMO controls it is important to look for these 2 affects. For example it is important to check how much the fluorophore in question affects the resolution of all channels it may spill into, as well as determining whether other fluorophores affect the channel in question. Plotting all fluorophores against each other with N X N plots and then overlaying all FMOs is a good way to determine these affects and make well-informed changes as required.
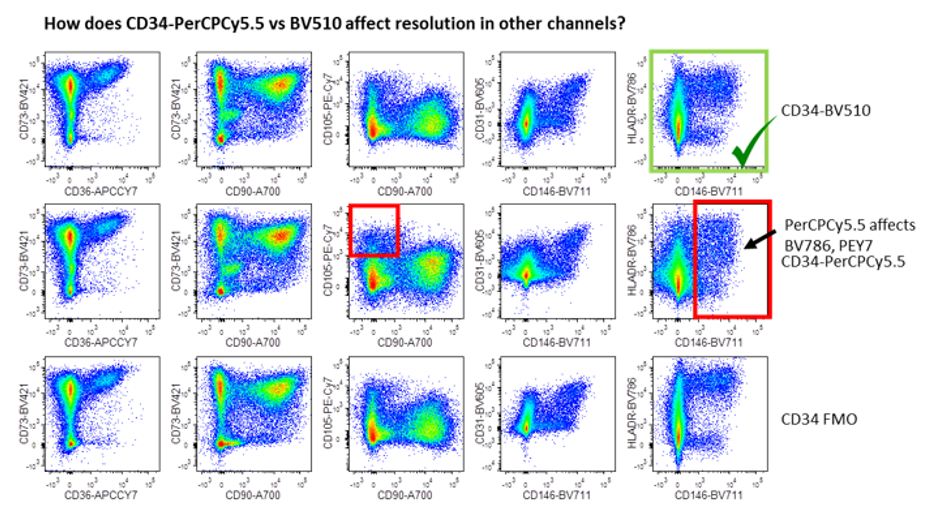
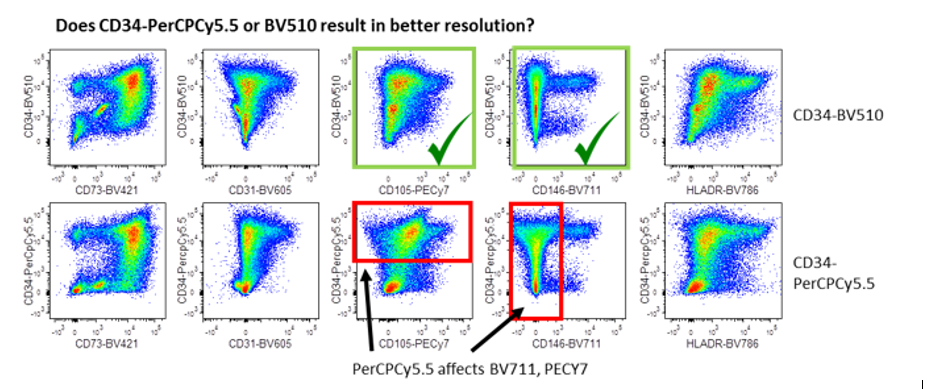
This figure demonstrates the 2 ways FMOs can reveal spillover problems. The first (top panels) demonstrates how the dye in question (either PerCPCY5.5 or BV510), affect the resolution in other channels by comparing to the FMO. The red boxes highlight areas where PerCPCY5.5 affected resolution (eg PECY7 and BV786). The green box shows BV510 is the better choice. The second panel reveals which of these dyes gives a better resolution of the antigen in question, i.e. CD34. PerCPCY5.5 causes spreading or spillover into neighbouring channels (red boxes) when compared with BV510 (green boxes). Image courtesy of Dr Anna Brooks.
Analysis controls are those controls that help us gate data. Most of these controls have already been discussed in the context of experimental design, but many of these actually come into play at the analysis phase to help validate our gating decisions.
Autofluorescence is the term used to describe the natural fluorescence that occurs in cells. The most commonly observed autofluoresing molecules are NADPH and flavins. All cells will have some level of inherent fluorescence, however larger cells and more granular cells will have the most. Common cell types include, tumour cells, fibroblasts, stromal cells and tissue macrophages. Therefore we need to be aware of this phenomenon, especially when working with digested tissues, as cell mixtures containing cell types with different levels of autofluorescence can make data analysis and interpretation difficult. Therefore an unstained control will help reveal whether your sample contains a mix of autofluorescent cells so that strategies can be developed to manage this. Specifically, autofluorescence can result in an increase in background (width of the negative) which could diminish resolution of dim, specific signals. This therefore may have an impact on reagent choice for these cells with high inherent fluorescence.
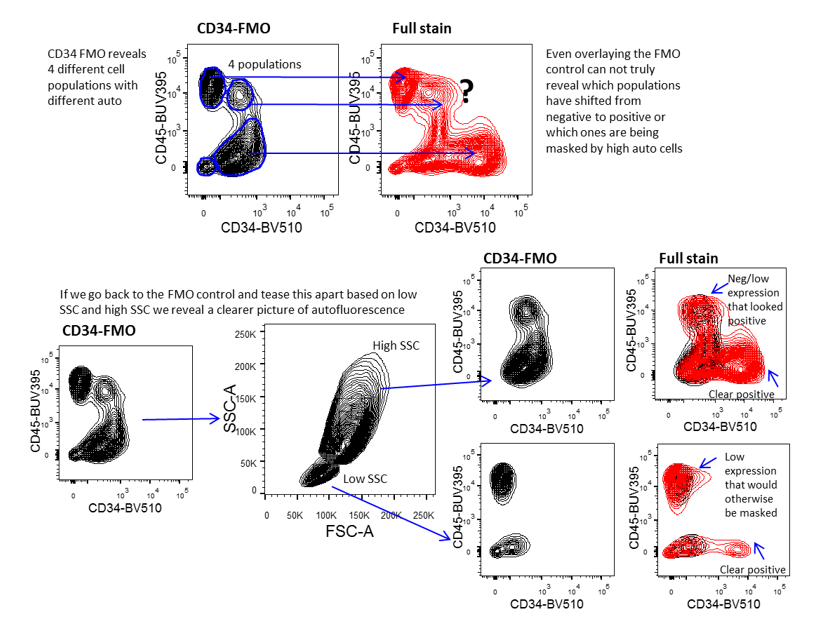
Use of FMO for gating control to manage autofluorescence in complex samples. This figure demonstrates the importance of using FMO controls to accurately gate data, especially when the samples contain cells with different levels of autofluorescence. Accurate analysis depends on teasing apart the different cell populations so that each cell population is assessed independently.
Using sufficient and correct controls can be the make or break of any scientific experiment. This is especially true in flow cytometry where there are many stages where error and variability can be introduced. Controls are required to ensure our instruments are performing well, reagents are staining as expected, fluorescent signals can be deconvoluted, positive (and negative) measurements are valid, and observations are legitimate and scientifically sound. All of these controls are equally important and critically required to ensure accurate interpretation of results.
Dr Anna Brooks holds a BCA (management) and a PhD in Immunology and is a Senior Research Fellow with the Maurice Wilkins Centre at the University of Auckland. Anna is also an expert flow cytometrist and is director of Auckland Cytometry, the flow cytometry core facility for the Faculty of Science. Anna’s primary interest lies in developing multicolour panels to characterise complex cellular populations in digested human tissues. Anna is also an active member of the international flow cytometry community and currently sits on the Australasian Cytometry Society Council as research liaison. When not marvelling in the many fluorescent colours of the cytometry world, Anna enjoys to scuba dive and explore the vibrant wonders of our underwater world.
If you enjoyed reading our articles, why not sign up to our blog mailing list? You'll get new articles straight to your inbox as they're released!